 
The main thing is that rusting involves the initial formation of Fe 2+ ions as the iron atoms lose electrons: All I can do here is to give a hint of the main processes, but without detail. This is surprisingly complicated, and to understand it properly, you will need to do chemistry for a few more years. The photo shows the effect of long exposure to sea water on a steel boat. Rusting is accelerated by the presence of dissolved ionic substances like salt. You need the presence of both air and water. It is a bit slow to start with, but is clearer than anything else I have been able to find.Īnd this short bit of time-lapse photography shows a steel plate rusting under more everyday conditions. The video below gives you all the information you want. So rather than protect the iron underneath, it still allows air and water to reach the iron. The problem with iron is that, unlike aluminium, the rust layer is very porous and flaky. 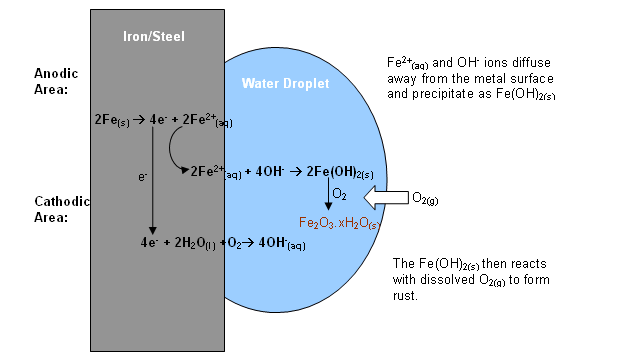
"x" shows a variable amount of water attached to the iron(III) oxide. Rust can be thought of as a hydrated iron(III) oxide, Fe 2O 3.xH 2O. The very strong, very thin, layer of aluminium oxide on the surface of aluminium helps to protect the aluminium. Many metals react with oxygen to form a layer of metal oxide on the surface. Rusting is the special name given to the corrosion of iron. You will need to understand the page about oxidation and reduction in the Reactivity Series section in terms of electron transfer.Ĭorrosion happens on the surface of a metal when the metal atoms react to form compounds such as oxides or carbonates or more complicated substances. This page looks at the rusting of iron, and the various ways it can be prevented or slowed down. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
